-
-
-
-
-
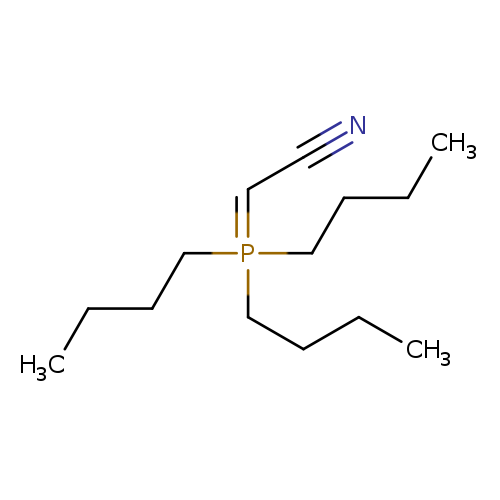
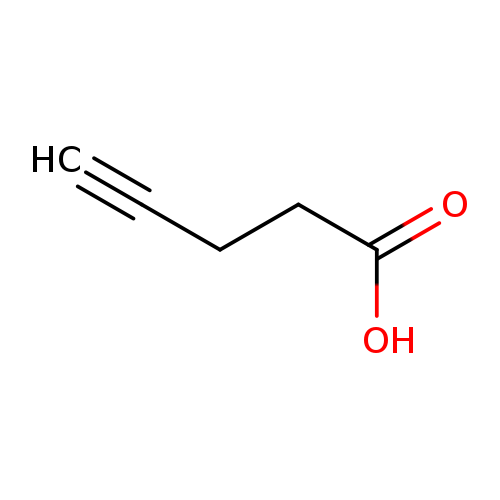
1P001PPC
157141-27-0 MFCD01321151
Acetonitrile, 2-(tributylphosphoranylidene)-
Pricing & Availability -
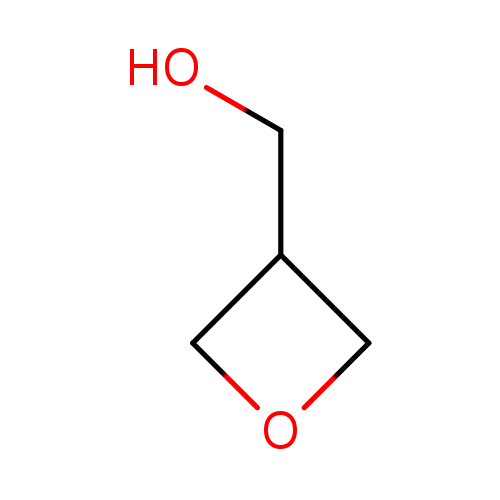
-
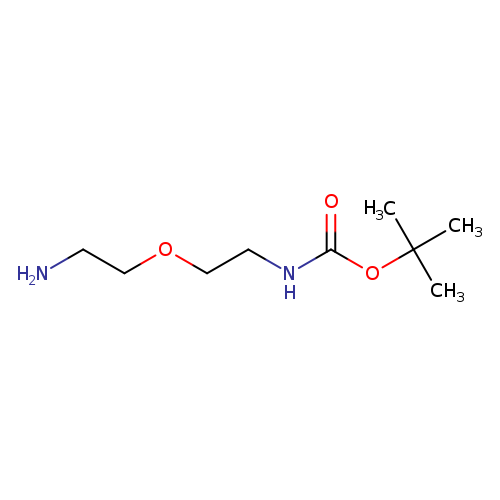
1P000XX7
127828-22-2 MFCD12031501
Carbamic acid, N-[2-(2-aminoethoxy)ethyl]-, 1,1-dimethylethyl ester
Pricing & Availability -
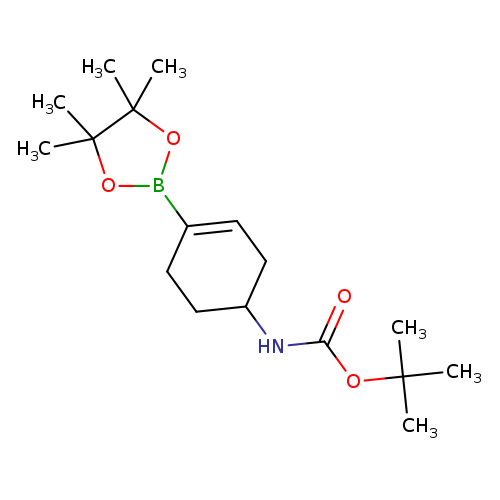
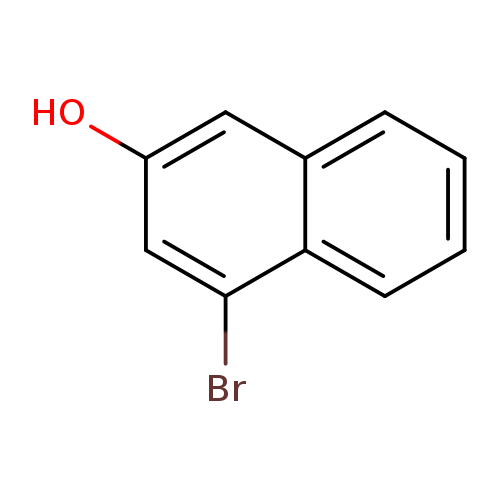
1P000NR8
1251732-64-5 MFCD12032216
Carbamic acid, N-[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-3-cyclohexen-1-yl]-, 1,1-dimethylethyl ester
Pricing & Availability -
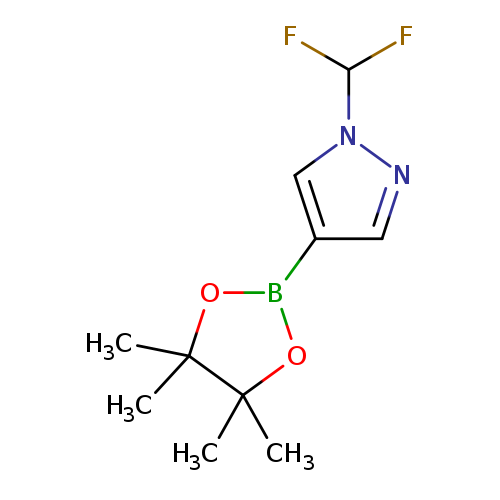
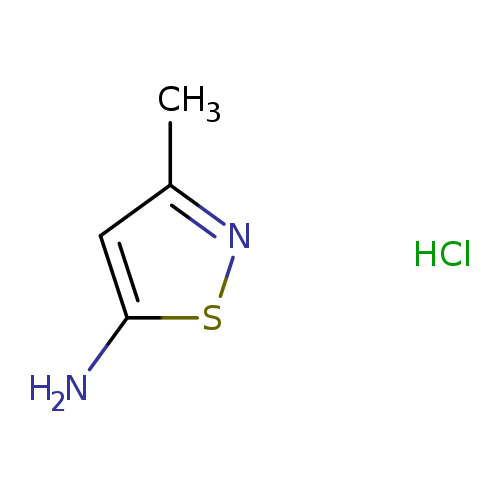
1P007Q5G
1206640-82-5 MFCD17011852
1-(Difluoromethyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole
Pricing & Availability -
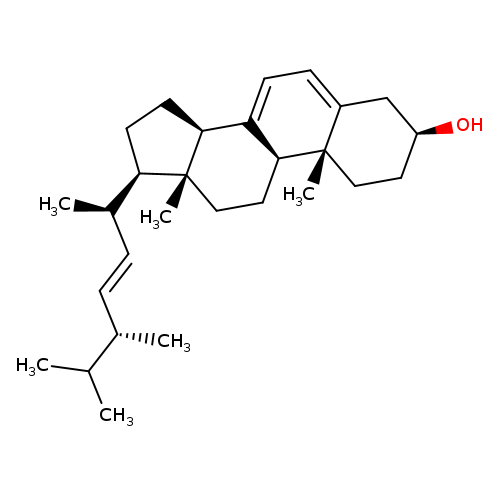
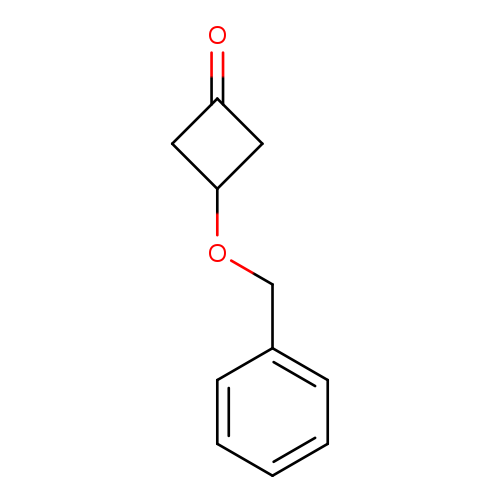
Ergosta-5,7,22-trien-3-ol, (3b,22E)-
Catalog#:1P00IAMY
CAS:57-87-4
MDL:MFCD09953680
Molecular Formula:C28H44O
Molecular Weight:396.6484
UN Number:
Haz Class:
Packing Group:
Keywords: 57-87-4,MFCD09953680,1P00IAMY,Ergosta-5,7,22-trien-3-ol, (3b,22E)-,C28H44O
Inquire| Catalog# | Purity | Size | Price | Dis. Price | Availability | Quantity | ||
|---|---|---|---|---|---|---|---|---|
| 1P00IAMY | 95% | 1g | $63.00 | $48.00 | In Stock USA | ADD TO CART | BUY NOW | |
| 1P00IAMY | 95% | 5g | $127.00 | $95.00 | In Stock USA | ADD TO CART | BUY NOW | |
| 1P00IAMY | 95% | 25g | $413.00 | $310.00 | In Stock USA | ADD TO CART | BUY NOW | |
| 1P00IAMY | 95% | 100g | $886.00 | $665.00 | In Stock USA | ADD TO CART | BUY NOW |
